It’s ‘FDA Approved’? Here’s what that really means.
The U.S Food and Drug Administration (FDA) does important work. The Administration is responsible for protecting public health in the U.S by regulating “human drugs and biologics, animal drugs, medical devices, tobacco products, food (including animal food), cosmetics, and electronic products that emit radiation”.
Simply put, they strive to keep the public safe. As we all know, they try to ensure that products Americans put into their bodies really are what they claim to be.
But a recent post on the Administration’s website points out some facts about how it all works. And some of it might come as a surprise.
(photo credit: www.pexels.com)
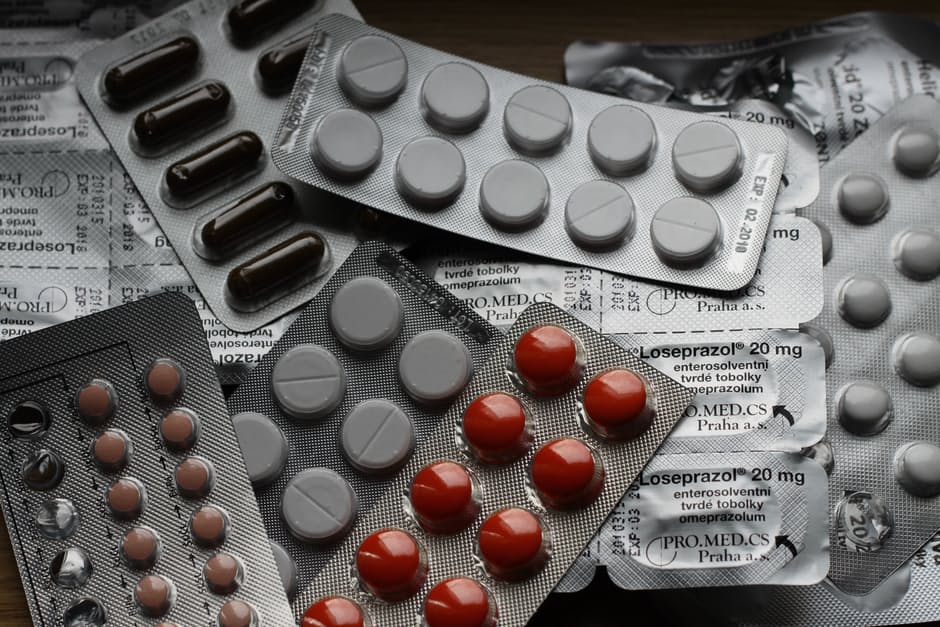
How many of us have searched for ‘FDA Approved’ labels on products, at the very least, for some peace of mind? And it makes sense to do so, because doesn’t the FDA test products to make sure they’re safe?
Actually, it turns out, no. The FDA doesn’t test anything. It’s all up to the manufacturers. And the Administration doesn’t actually evaluate these, either.
What the FDA does is simply review the results of testing that’s been done by the manufacturer, the Administration explains.
(photo credit: www.pexels.com)
FDA experts look at the test results that manufacturers provide them with, and with these the Administration determines if they’ll approve a product to be safe enough, or not.
“If FDA grants an approval,” the Administration’s website states, “it means the agency has determined that the benefits of the product outweigh the known risks for the intended use.”
Related: The First Artificial Pancreas Has Been Approved by the FDA
It sounds like a great statement. But then when you think about it, all the FDA approved pharmaceutical drugs out there come to mind. And then the mile-long lists of side effects that accompany them. What’s up with all that? Would everyone agree that the benefit outweighs the risk of taking them, in all cases?
Manufacturers and Laboratories
(photo credit: www.pexels.com)
Anyways, back to concrete things, like factories.
So, how can you, the consumer, know if a manufacturer, healthcare facility or laboratory is following the rules? You can’t, really. You just have to hope that it’s honest. The public can cross its fingers and hope that the place is honorable and that maybe it’s been looked at by the right people.
Related: No Baby Products on the Market Will Prevent SIDS: FDA
The Administration simply has the authority to inspect regulated facilities to make sure they are up to code with manufacturing practice regulations. And they only do so if they choose to.
The results are manifold. The FDA openly admits that their enforcement efforts sometimes don’t get to all products before they come to market. Sometimes, it happens after a product has already hit the shelves.
(photo credit: www.pexels.com)
What does this mean for consumers? Well, in some cases a product could potentially seem safe, but maybe not necessarily be so. And there could be other ramifications if you care to think about them, but that’s a rant for another time.
Related: What is ‘Healthy Food’? The FDA Turns Their Most Popular Query to the Public
If you’re concerned about something you’ve bought or are taking, talk to your doctor.
Consumers can also contact the Administration at 1-888-INFO-FDA (1-888-463-6332), by clicking here, or by mailing an inquiry to:
Food and Drug Administration
10903 New Hampshire Ave
Silver Spring, MD 20993-0002
Photo credit: Castleski/Shutterstock