In a scientific discovery that could be life-changing for people with spinal injuries, a primate regained control of a paralyzed limb thanks to a new brain implant.
The implant wirelessly restores the broken communication between the body’s brain and spinal cord.
“This is the first time that you have a connection between the brain and the spinal cord that re-established movements,” the study’s leader, Grégoire Courtine, a neuroscientist at the École Polytechnique Federale de Lausanne (EPFL) in Switzerland, told CBC News.
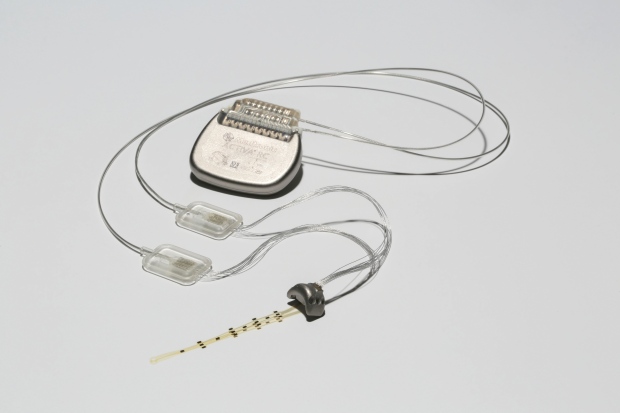
Scientists have utilized wireless implants to return movement before, though serious, noticeable hardware needed to compliment it, like a prosthetic limb or mechanical exoskeleton. This stunning brain-spine interface, however, employs just two surgical implants to wirelessly relay information.
How Does It Work?
A normally functioning nervous system works through a region of the brain called the motor cortex. The cortex sends signals –think of them as instructions for the body – through the spine, reaching a neural network in the lumbar region. From there, the neurons interpret the instructions, and activate the appropriate muscles, thus facilitating movement.
A spinal cord injury stops these signals from reaching its lumbar destination.

Using the brain-spine interface, the implant picks up the brain activity, sending it to a computer that decodes the signals. Those instructions are relayed to a spinal cord simulator, which is embedded in the paralyzed person’s lumbar region.
Related: Paralyzed man moves legs using brain-reading device
“The link between the decoding of the brain and the stimulation of the spinal cord — to make this communication exist – is completely new,” neurosurgeon Jocelyne Bloch of the Lausanne University Hospital, who surgically affixed the implants, said in a release.
“For the first time, I can imagine a completely paralyzed patient able to move their legs through this brain-spine interface.”
Will it Work on Humans?
It’s still unclear whether this technology will work on humans, especially those with severe spinal cord damage; the technology has only been tested on primates thus far. While the researchers of the study are encouraged by their clinical trial, they did acknowledge the monkey in the study had a small lesion, and would’ve eventually regained its motor functions on its own eventually.
“You need to be realistic,” Courtine said. “We don’t imagine that tomorrow there will be paralyzed people who will walk in the streets with a brain-spine interface. We are not there yet.”
While Courtine’s team continues to test the interface, he says the technology can still provide value outside its intended purpose. It’s said to have practical applications for rehabilitating injured patients; in physical therapy, a patient has to re-learn how to walk, but with this implant, they can potentially walk right away. The monkey in the study didn’t need any rehab – it immediately found its legs, so to speak.
“The key is there’s no rehabilitation; there’s no training on the animal,” Courtine said.
There’s already another study underway that’s evaluating the interface’s therapeutic potential of the spinal cord stimulation technology, without the brain implant. It may be able to improve the walking of people with only partial spinal cord injuries.
The research from EPFL, who collaborated with Medtronic, Brown University and Fraunhofer ICT-IMM on the project, was published yesterday in the journal Nature.












